+86-18343147735
+86-18343147735
0102030405
News
Recent developments in the EU IVD regulatory landscape
2025-03-07
In vitro diagnostics (IVDs) have become a cornerstone of modern medicine, and rising demand for these critical medical devices has driven rapid market growth. Since 2021, hospitals and laboratories...
view
detail

The In Vitro Diagnostics (IVD) market is undergoing significant transformation
2025-03-07
The In Vitro Diagnostics (IVD) market is undergoing significant transformation, driven by technological advancements, shifting market dynamics, and evolving regulatory landscapes. As we navigate th...
view
detail

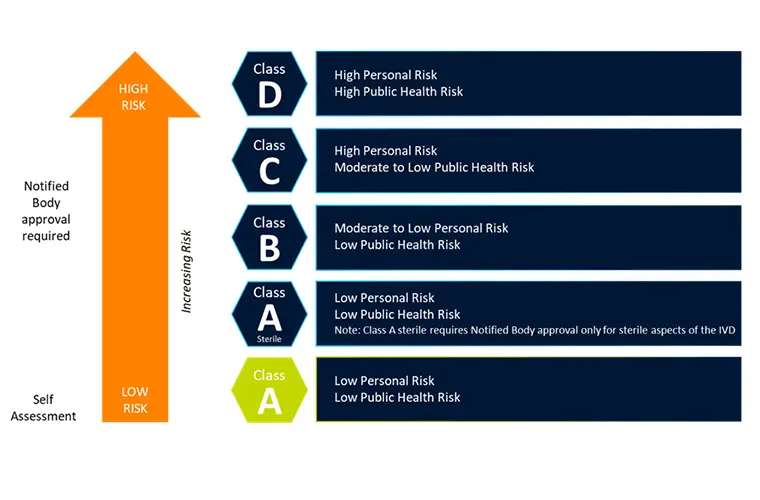
In Vitro Diagnostic Regulation (IVDR)
2025-03-07
The In Vitro Diagnostic Regulation (IVDR) replaced the IVDD and entered into force on 26 May 2017 with 26 May 2022 as date of applicationIn March 2023, the IVDR was amended as regards to transition...
view
detail