+86-18343147735
+86-18343147735
0102030405
News

WHX Miami 2025 — A Global Hub for Medical Device Innovation & Trade
2025-06-16
Here’s a polished English news-style summary of WHX Miami 2025 (formerly FIME) with key highlights and context:
Event Overview
WHX Miami 2025, the rebranded Florida International Medical E...
view
detail

FDA Accelerates Home Test Approvals: POCT Is the Future
2025-06-11
1:Policy Shift: Home Diagnostics Enter the Fast Lane
view
detail
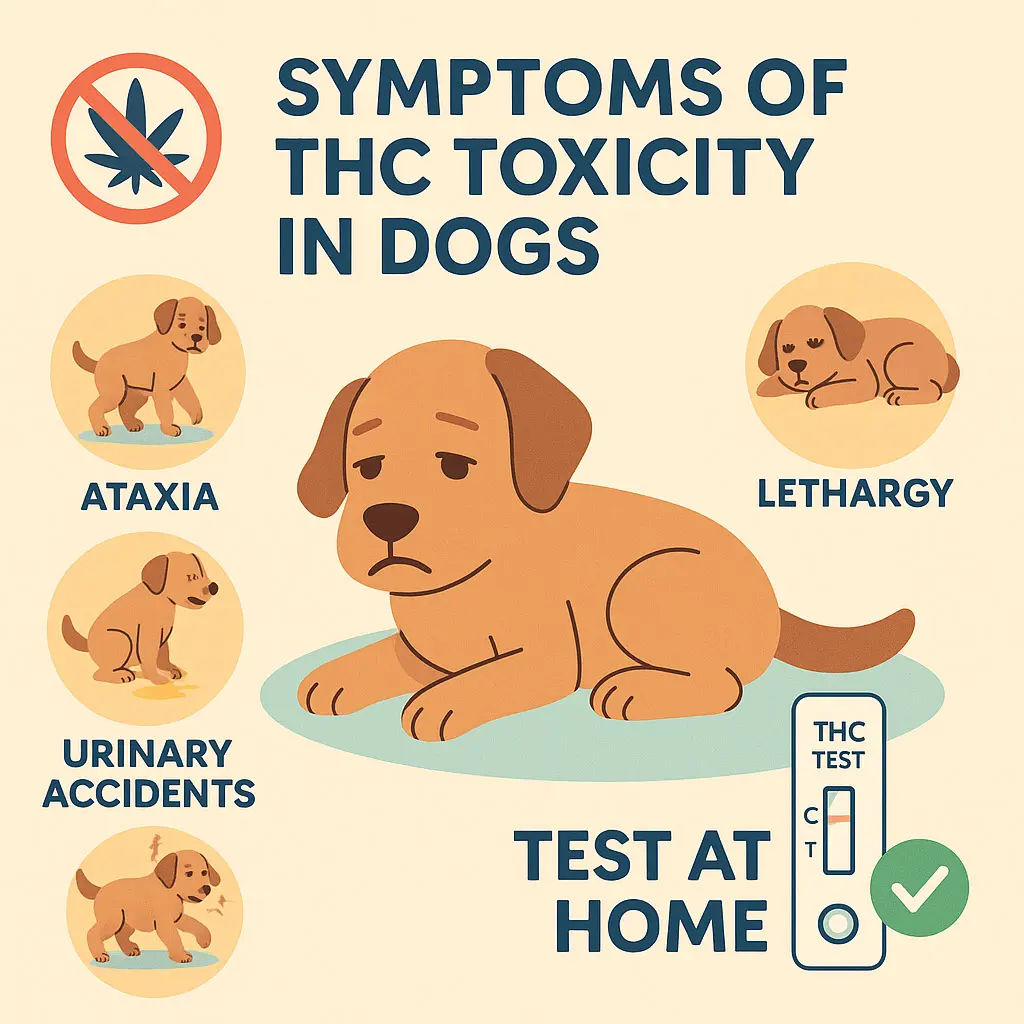
THC in Pets? What Most Owners Don’t Realize
2025-06-05
Unseen Risks, Subtle Symptoms, and the Need for At-Home Testing
view
detail

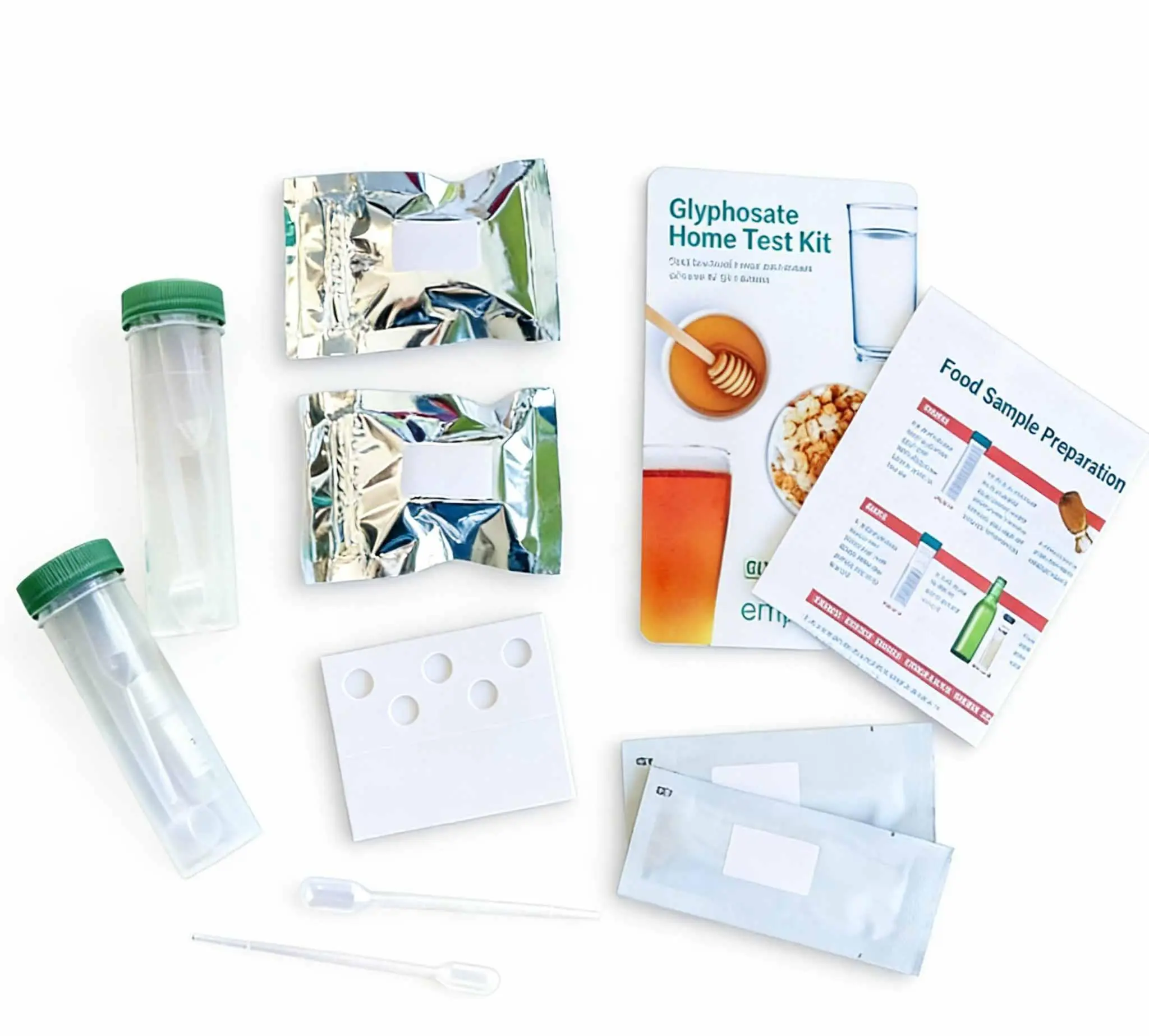
Understand in 10 Minutes: Home Test vs. Lab Test — What Really Sets Them Apart
2025-06-11
Many assume home testing is “less accurate” or “unprofessional.” In fact, certified home-use kits rely on the same biological principles as lab-based tests—only the sc...
view
detail

FDA-Cleared 12-Panel At-Home Drug Test Kit Empowers Families and Workplaces with Rapid Screening
2025-06-01
Conlight Diagnostics launches an FDA 510(k)-cleared immunoassay urine cup test, enabling self-administered screening for 12 common drugs of abuse in under 10 minutes.
Shanghai, China – June ...
view
detail

The In Vitro Diagnostic Regulation (IVDR) replaced the IVDD and entered into force on 26 May 2017 with 26 May 2022 as date of application
2025-05-16
In March 2023, the IVDR was amended as regards to transitional provisions for certain in vitro diagnostic (IVD) medical devices on Article 110(4) with the removal of the sell-off period to prevent ...
view
detail

Key changes introduced by IVDR
2025-05-16
In vitro diagnostics (IVDs) have become a cornerstone of modern medicine, and rising demand for these critical medical devices has driven rapid market growth. Since 2021, hospitals and laboratories...
view
detail
Future Drivers and Challenges
2025-05-15
The In Vitro Diagnostics (IVD) market is undergoing significant transformation, driven by technological advancements, shifting market dynamics, and evolving regulatory landscapes. As we navigate th...
view
detail

The Shift from IVDD to IVDR—and the Challenges
2025-05-08
The shift from IVDD to IVDR represents a major change in how the IVD industry is regulated. While the EU Commission maintains that the system’s overall structure and approach remain consist...
view
detail