+86-18343147735
+86-18343147735




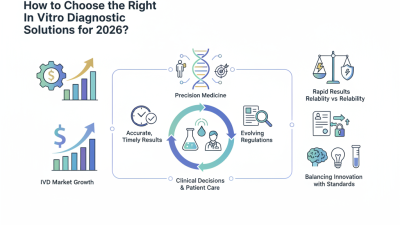
In the evolving field of In Vitro Diagnostic (IVD) solutions, significant advancements are seen globally. Dr. Emily Chen, a prominent expert in the IVD sector, states, “Innovations in diagnostics can transform patient care and treatment outcomes.” This highlights the importance of IVD technologies for improving healthcare worldwide.
The global market for In Vitro Diagnostic solutions continues to expand rapidly. Buyers are seeking reliable, efficient products to meet growing demands. The challenge lies in evaluating options that ensure accuracy while addressing cost-effectiveness. In a field with diverse offerings, pinpointing the right solutions requires expertise and understanding of specific needs.
Though many solutions promise high performance, not all deliver on that promise. Stakeholders must remain vigilant, questioning the reliability of various products. Engaging with industry experts can provide valuable insights. The pursuit of quality in In Vitro Diagnostic solutions is essential, yet it often requires ongoing assessment and critical reflection.

In vitro diagnostic (IVD) solutions are essential in today’s healthcare landscape. These products are used to analyze samples outside the body. They help in detecting diseases, monitoring health, and guiding treatment decisions. The global IVD market has seen remarkable growth over the years, driven by technological advancements and increasing healthcare needs.
Tips: When considering IVD solutions, focus on accuracy and reliability. Choosing products that have undergone rigorous testing is crucial. Pay attention to regulatory approvals in various regions to ensure compliance.
Many buyers may overlook the importance of local needs. Market demands can vary significantly across regions. Understanding these nuances can enhance the effectiveness of diagnostic solutions. Always consider adapting solutions to fit specific healthcare environments.
Continuous innovation shapes the future of IVD. However, not all advancements prove beneficial immediately. Some technologies face challenges during implementation. Regular evaluation of new products can help identify potential issues early on.
The in vitro diagnostic (IVD) industry is rapidly evolving. Recent reports highlight that the global IVD market is projected to reach over $85 billion by 2025, driven by increasing healthcare demands. Key players are focusing on innovative solutions tailored to specific diseases, aiming to enhance diagnostic accuracy and patient care. The reliance on advanced technologies, like molecular diagnostics, is transforming traditional practices.
Major companies in the IVD sector invest significantly in research and development. This investment leads to breakthroughs in detecting diseases at early stages. According to industry analyses, the rise of point-of-care testing is influencing purchasing decisions globally. However, challenges persist, including regulatory hurdles and the need for significant clinical validation of new tests.
Collaboration between firms and research institutions is becoming essential. This synergy enables sharing of insights and accelerates product development. Despite a competitive landscape, there is still room for improvement. Many players struggle with adapting to rapid technological changes and meeting evolving patient needs. The industry must reflect on these challenges to ensure sustainable growth and enhance diagnostic solutions.
| Diagnostic Solution | Market Segment | Key Features | Growth Rate (%) |
|---|---|---|---|
| Polymerase Chain Reaction (PCR) | Molecular Diagnostics | High sensitivity and specificity | 8.5% |
| Enzyme-Linked Immunosorbent Assay (ELISA) | Immunodiagnostics | Versatile and widely used | 6.2% |
| Rapid Tests | Point-of-Care Testing | Quick results, easy to use | 10.1% |
| Next-Generation Sequencing (NGS) | Genomics | High throughput and accuracy | 15.3% |
| Microbiological Culture | Microbiology | Standard method for pathogen detection | 4.8% |
Emerging technologies are revolutionizing the field of in vitro diagnostics. According to a recent report by Research and Markets, the global in vitro diagnostics market is projected to reach $93 billion by 2025. This growth is primarily driven by advancements in molecular diagnostics and point-of-care testing.
Molecular diagnostic techniques, such as PCR and next-generation sequencing, offer unprecedented accuracy. These technologies facilitate rapid disease detection, crucial in managing outbreaks. A study highlighted that molecular testing can reduce the time to diagnosis by up to 50%. This not only improves patient outcomes but also enhances public health responses.
Moreover, point-of-care testing has gained traction in clinical settings. Devices are becoming smaller and more user-friendly. A report by IBISWorld indicates that the point-of-care market is growing at an annual rate of 11%. However, there are areas needing attention. Reliability can fluctuate; some devices may not perform well in diverse conditions. Addressing these gaps is vital for future advancements in diagnostics.

When buying in vitro diagnostic solutions globally, understanding regulatory frameworks is crucial. Each region has specific requirements that influence the approval and sales of diagnostic products. Buyers must familiarize themselves with local regulations and standards. This knowledge ensures compliance and product effectiveness.
Navigating these regulations can be challenging. Some markets may demand extensive documentation and rigorous testing before approval. This leads to delays and increased costs. Thus, it is vital for buyers to stay updated on regulatory changes. Regular training and consultation with regulatory experts can aid in this process.
In addition to regulatory hurdles, buyers must consider the quality assurance of diagnostic solutions. Quality indicators vary across different regions. Understanding what constitutes quality can be overwhelming. Establishing a checklist based on regulatory best practices can mitigate risks. A clear focus on these criteria can enhance the buyer's decision-making process.

The in vitro diagnostics (IVD) market is rapidly evolving, influenced by various trends. A recent report predicts the market will reach approximately $100 billion by 2025. This growth emphasizes the increasing need for advanced diagnostic solutions. Key areas driving this change include technological advancements and the rise of personalized medicine.
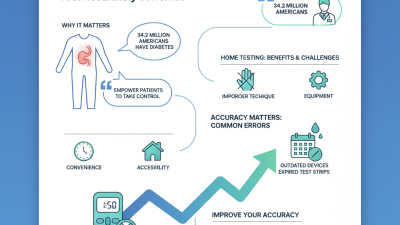
Artificial intelligence (AI) is becoming integral to IVD. AI-enhanced tools improve accuracy and speed in disease detection. For example, they can reduce diagnostic errors by up to 30%. Furthermore, point-of-care testing is gaining traction. This provides immediate results, crucial for timely treatment decisions. A significant shift toward home-testing kits also indicates how consumer preferences are reshaping the industry.
However, challenges remain. Regulatory hurdles often slow down innovation. Ensuring data privacy is another pressing concern. As the market grows, so does the need for stringent regulations. Balancing innovation with compliance will be vital. Stakeholders must navigate these issues to fully harness IVD's potential. The future will depend on how effectively these trends are managed.