+86-18343147735
+86-18343147735




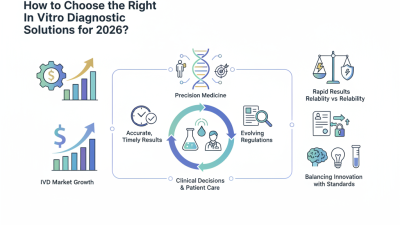
Choosing the right In Vitro Diagnostic tools is critical for any laboratory. A well-informed decision can enhance accuracy and efficiency in testing. Each laboratory has unique needs influenced by the tests they perform and the patient populations they serve.
In Vitro Diagnostic technology has evolved rapidly. New tools can provide faster results and improved accuracy. However, selecting the right equipment is not a straightforward process. Laboratories must consider factors such as budget, space, and intended use. Understanding the performance and reliability of these tools is essential. The wrong choice can lead to misdiagnoses or workflow inefficiencies.
With numerous options available, it's easy to feel overwhelmed. Laboratories must evaluate each tool’s features and the manufacturer’s reputation. Engaging with peers and reading reviews can provide valuable insights. Reflecting on past experiences with diagnostic tools may also help guide future choices. Ultimately, the right In Vitro Diagnostic tools are those that align best with the laboratory's goals and patient needs.

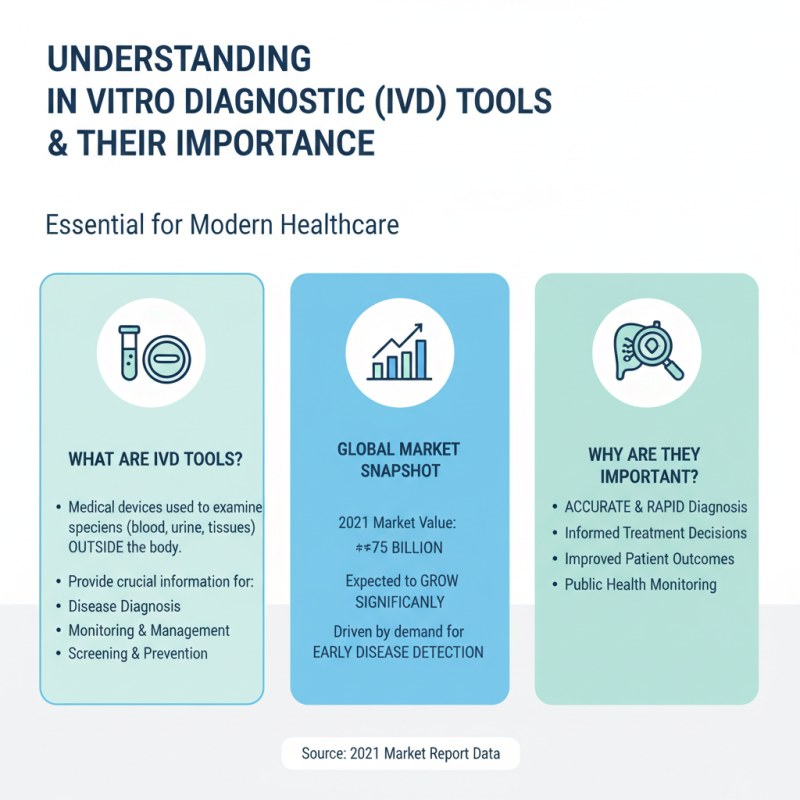
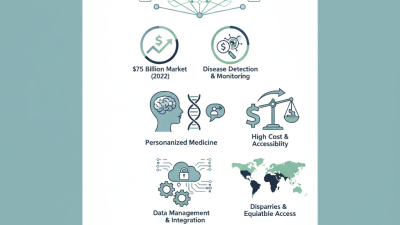
In vitro diagnostic (IVD) tools are essential for modern laboratories. They enable accurate disease diagnosis and management. According to a recent market report, the global IVD market was valued at approximately $75 billion in 2021 and is expected to grow significantly in the coming years. This growth reflects the rising demand for early disease detection.
Choosing the right IVD tools requires a deep understanding of their applications and functionalities. Laboratories often face challenges in selecting the most suitable instruments. For instance, regulatory compliance is crucial. The average delay in obtaining regulatory approval can extend laboratory workflows. Moreover, facilities may struggle with integrating new technologies into their existing systems. A 2022 survey indicated that 25% of laboratories found integration to be a significant obstacle.
Despite the advances in IVD tools, several issues still need addressing. The complexity of certain diagnostics can lead to user error. Technicians may require thorough training to minimize these risks. Additionally, data management remains a critical concern. With increasing data volume, laboratories may find it difficult to ensure accuracy and reliability in results. Continuous improvement and adaptation are necessary for maximizing the potential of IVD tools.
Identifying the needs and requirements for diagnostic tools is a critical step in optimizing laboratory performance. Begin by assessing the specific tests your laboratory aims to conduct. Understand the types of samples you will process and the expected turnaround times. This foundational knowledge will help you narrow down the necessary features for your in vitro diagnostic tools.
Consider the regulatory landscape as well. Compliance with relevant standards is essential for avoiding issues down the line. Analyzing your current diagnostic capabilities can also highlight areas needing improvement. Are there gaps in the types of tests offered or delays in processing? Reflecting on these aspects may reveal the need for more advanced technologies or user-friendly interfaces.
Engage with your laboratory staff to gather insights as well. They can provide feedback on existing tools and suggest improvements based on their experiences. This collaborative approach fosters a work environment where everyone feels valued. Feedback can lead to better-informed decisions when selecting new tools. Aim for a balance between technical specifications and ease of use, ensuring the diagnostic tools truly fit your laboratory's needs.
When selecting in vitro diagnostic (IVD) tools, understanding the different technologies is crucial. Many laboratories deal with various types, including molecular, immunoassay, and biochemical methods. Each has unique strengths and weaknesses impacting accuracy and speed. For example, molecular diagnostics offer high specificity but require thorough training. In contrast, immunoassays can provide quick results but may deliver less precise outcomes.
It’s vital to consider your laboratory's specific needs. Some environments prioritize rapid testing, while others require deep analytical capabilities. Assessing the test volume will also influence your decision. Too many options without clear guidance can lead to confusion. Gathering input from experienced lab professionals can provide valuable insights.
Another essential factor is user-friendliness. Complex systems may require extensive training. If a technology is overly complicated, it can hinder daily operations. Reflecting on past decisions can help navigate future choices better. Balancing innovation with practical application is key to making informed selections.

When selecting in vitro diagnostic tools, quality and accuracy are paramount. Not all diagnostic tools offer the same reliability. Variability in manufacturing can lead to differences in performance. Laboratories must assess the specific needs for their tests. Understanding the precision and sensitivity of each tool is crucial.
Using independent reviews and performance data provides insight into various brands. Some diagnostic tools may excel in certain areas but underperform in others. Investigating feedback from lab professionals can reveal hidden strengths and weaknesses. A tool that is popular may not always be the best choice for your lab's specific requirements.
Moreover, regular assessments of tool performance can identify areas for improvement. Tracking accuracy rates over time offers valuable feedback. It’s essential to maintain an adaptable mindset. The best diagnostic tools today may not be the best tomorrow. Continuous education and research play a vital role in keeping laboratories equipped with the right tools.
Choosing the right in vitro diagnostic tools hinges heavily on understanding regulatory compliance. Regulatory bodies, like the FDA, set strict guidelines. Products must meet specific safety and efficacy standards. The premarket approval process can be rigorous. It ensures that diagnostic tools are reliable and effective.
Some studies indicate that nearly 40% of diagnostic devices fail to meet regulatory requirements on first submission. Laboratories must stay informed about updates in regulations. Legislative nuances can change frequently. This is where expertise plays a crucial role. Engaging professionals with knowledge of the regulatory landscape can mitigate risks.
Certification is another essential aspect. It often serves as evidence of quality and reliability. Tools that are CE-marked or ISO-certified typically demonstrate a higher level of compliance. However, not all certifications are equal. A tool may be certified but still need performance assessment. Therefore, ongoing evaluation of tools is vital post-certification. This practice helps in maintaining high standards in laboratory results.
| Diagnostic Tool Type | Regulatory Compliance Standard | Certification Body | Approval Status | Average Cost (USD) |
|---|---|---|---|---|
| PCR Kits | CLIA | FDA | Approved | $150 |
| ELISA Kits | ISO 13485 | CE | Approved | $120 |
| Immunoassays | FDA 510(k) | FDA | Pending | $200 |
| Rapid Tests | CLIA | WHO | Approved | $50 |
| Microbial Culture Tests | ISO 15189 | FDA | Approved | $75 |