+86-18343147735
+86-18343147735




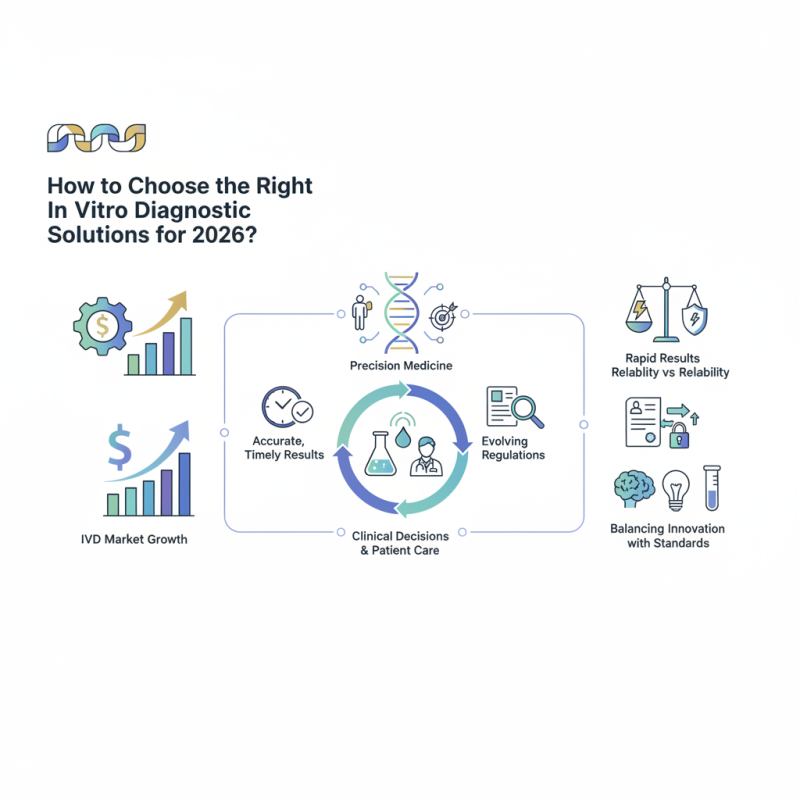
The landscape of In Vitro Diagnostic (IVD) solutions is evolving rapidly. As we look toward 2026, understanding the key trends in this field is crucial. The global IVD market is projected to reach $87 billion by 2026, with a compound annual growth rate of 4.7% (Frost & Sullivan). This growth underscores the importance of selecting the right diagnostic tools.
In the coming years, precision medicine will take center stage. The demand for personalized diagnostics is increasing, as healthcare shifts towards tailored treatments. This shift requires diagnostics that provide accurate, timely results. Data indicates that 70% of clinical decisions are based on laboratory results (Clinical Laboratory Improvement Amendments). Thus, choosing appropriate IVD solutions is not just essential but critical for patient care.
However, the selection process is not without challenges. Many products claim rapid results but may lack reliability. Additionally, evolving regulations can complicate decision-making. Balancing innovation with established standards requires thoughtful consideration. In conclusion, navigating the IVD landscape will require careful analysis of emerging trends and technologies. A well-informed choice can significantly impact healthcare outcomes.

As we look ahead to 2026, the landscape of in vitro diagnostics (IVD) is continuously evolving. Advances in technology play a significant role in shaping these solutions. The integration of artificial intelligence in diagnostics will likely streamline processes and enhance accuracy. Yet, the challenge remains in ensuring these tools are user-friendly and accessible to a wider audience.
Regulatory changes are expected to influence the IVD market. Stricter regulations may lead to improvements in product safety and efficacy. However, adapting to these changes could create hurdles for smaller companies. They might struggle with compliance, potentially stifling innovation. It’s crucial to strike a balance between regulation and innovation to foster growth.
Moreover, the ongoing need for personalized medicine demands robust diagnostic solutions. Precision diagnostics must evolve alongside this trend, focusing on individual patient needs. Yet, the complexity of data interpretation can be daunting. Ensuring that healthcare professionals have the necessary skills to manage this data is essential. Ignoring this could lead to misinterpretation and delivery of inadequate care.
As we approach 2026, navigating the landscape of in vitro diagnostics (IVDs) requires careful consideration of regulatory changes. Recent shifts in regulations aim to ensure safety and efficacy. These updates can significantly impact how diagnostic solutions are developed and presented.
Regulatory bodies are now prioritizing transparency and data quality. They expect companies to provide robust evidence of clinical validity. This is where challenges often arise. Many companies struggle to meet these new demands. They may need to invest more time and resources to gather necessary data, leading to delays and increased costs.
Innovation should not stagnate amid these regulatory challenges. Understanding the intricacies of regulatory changes can yield opportunities. Companies that adapt quickly will likely thrive. Yet, it’s essential to continuously evaluate the long-term implications of these regulations. Balancing compliance with innovation remains a critical, ongoing task.
Selecting the right in vitro diagnostic solutions for 2026 involves understanding the latest technological advancements. Diagnostic devices are rapidly evolving. They now offer enhanced accuracy and faster results. This evolution is crucial for timely patient care. Technologies like AI and machine learning are gaining traction. They help in pattern recognition and data analysis, boosting diagnostic efficiency.
**Tips:** Consider compatibility with existing systems. Evaluate ease of use for healthcare personnel. Check if the technology integrates well with other diagnostic tools.
Advancements should also be scrutinized. Not all new technologies are better. Some may require extensive training, slowing down adoption. Also, newer devices may have teething issues. Understanding these aspects is vital. Look for solutions that balance innovation with reliability. Safety and regulatory compliance should never be overlooked.
**Tips:** Stay updated on regulatory changes. Follow industry news for emerging technologies. Engage with professional networks for insights.
This bar chart illustrates the projected growth of the in vitro diagnostic solutions market from 2020 to 2026. It shows a steady increase in market size, reflecting the advancements in diagnostic technologies and growing demand in healthcare.
Understanding market demand for in vitro diagnostic solutions in 2026 requires keen observation. Consumers today are more informed and selective. They prioritize accuracy and reliability in test results. A focus on user-friendly solutions is also essential. Simplicity can lead to better adherence to testing protocols.
Environmental concerns will play a big role in choices. Eco-friendly solutions might appeal more to consumers. Innovations that reduce waste are gaining traction. However, not all consumers value these factors equally. Some may prioritize cost over environmental impact.
Demographics can influence preferences too. Younger consumers often seek convenience through digital solutions. Older populations might prefer traditional, familiar methods. Markets are diverse. Recognizing these nuances is vital. The need for adaptability in product offerings is clear. Current trends indicate that flexibility in response can improve market reach significantly.

In 2026, choosing the right in vitro diagnostic (IVD) solutions requires careful evaluation of cost-effectiveness. Recent studies indicate that the global IVD market is expected to reach $100 billion by 2026. With numerous products available, distinguishing between effective and economical options is crucial. Many providers claim their products offer detailed accuracy and faster results. Yet, the value often varies based on healthcare settings.
Cost-effectiveness analysis reveals that some leading IVD products can save up to 30% compared to traditional methods. However, this figure doesn’t always translate to better patient outcomes. A report notes that around 20% of diagnostic tests may not provide significant clinical utility. This means that health systems need to gauge not just the price, but actual benefits of each solution. In some cases, the initial low cost could lead to higher long-term expenses due to unnecessary follow-ups or misdiagnoses.
Moreover, integration with existing workflows remains a challenge. Many products claim seamless adoption, yet 40% of laboratories report difficulties in implementation. Such issues can negate any claimed advantages in cost or efficiency. It’s crucial to critically analyze these factors before making a selection. The balance between upfront costs and potential outcomes is often a gray area in the decision-making process.